Cryptogenic stroke has been a challenge for years in
clinical practice, given it can represent up to 40% of strokes,
depending on the etiological classification used, which has different
operative definitions for those patients in whom the complete diagnostic
workup does not reveal a specific stroke etiology. In 2014, two
opposing approaches to the challenge of cryptogenic stroke were
proposed. On the one hand, Bang et al. proposed its assessment using
advanced diagnostic techniques (such as advanced vascular imaging and
cardiac monitoring, aortogenic, and paradoxical embolic source
assessment, coagulopathy, and cancer screening tests) as a measure to
reduce the proportion of cryptogenic strokes by increasing the diagnosis
of atheroembolic disease, aortic embolic disease, branch occlusive
disease, paroxysmal atrial fibrillation, paradoxical embolism, and
cancer-related coagulopathy (1). On the other hand, the Cryptogenic Stroke/ESUS International Working Group proposed a new clinical construct that they named embolic stroke of undetermined source (ESUS) (2).
The rationale behind the ESUS concept was the assumption that most
cryptogenic strokes were thromboembolic and could benefit from
non-vitamin K agonist oral anticoagulants (NOACs) that had already
demonstrated their efficacy and safety in atrial fibrillation (AF), to
reduce recurrent brain ischemia. This suggestion prompted the
development of randomized clinical trials testing the safety and
efficacy of rivaroxaban and dabigatran in patients with ESUS as well as
the rapid dissemination of the ESUS concept to clinical practice due to
the simplicity of the diagnostic workup, which required only the
demonstration by neuroimaging procedures [computed tomography (CT) or
magnetic resonance imaging (MRI)] of a non-lacunar infarction, the
absence of extracranial or intracranial atherosclerosis causing ≥50%
luminal stenosis in arteries supplying the ischemic area, no major-risk
cardioembolic source of embolism and no other cause of stroke
identified. The only diagnostic procedure assessments required were
brain CT or MRI, 12-lead electrocardiogram (ECG), precordial
echocardiography, cardiac monitoring for ≥24 h with automated rhythm
detection and imaging of extracranial and intracranial arteries by
either ultrasonography or MRI, CT, or catheter angiography.
However, the failure of the RESPECT-ESUS and NAVIGATE-ESUS trials (3, 4)
in demonstrating any efficacy in the prevention of stroke recurrences
in patients with ESUS has called into question the practical usefulness
of this concept as, compared with the concept of cryptogenic stroke, the
only difference is the exclusion of lacunar stroke (5–9).
Therefore, ESUS remains a non-diagnosis similar to the classic
cryptogenic stroke concept. Some of the most commonly used stroke
classifications, such as the TOAST (10) and the ESUS criteria themselves (2),
were developed for use in clinical trials; however, they have been
incorporated in clinical practice without enough validation studies to
truly evaluate their usefulness in clinical settings. Stroke physicians
attend stroke patients daily in whom there is no clear evidence of the
underlying etiology and must choose between two options. One is to make
the diagnosis of ESUS (which is not actually associated with any
specific therapeutic change), and the other is to make a greater effort
to identify the ultimate cause of the stroke by the use of advanced
diagnostic techniques (1). Choosing one or the other option is crucial for secondary stroke prevention, given the treatments can be different.
In this opinion article, we would like to highlight some
of the arguments against the use of the ESUS concept in clinical
practice:
•
The rationale behind the ESUS concept, considering that many ESUSs
would be due to covert paroxysmal AF, and therefore would benefit from
the use of NOAC, has clearly failed, not only because of the
neutral/negative results of the RESPECT-ESUS and NAVIGATE-ESUS trials (3, 4),
but also because clinical trials with long-term ECG monitoring as well
as observational studies have shown that covert AF represents only about
30% of ESUS (11–13).
This
low rate of AF detection during follow-up, the different phenotypes
between patients with ESUS and patients with stroke with AF, and data
from studies with implantable cardiac monitoring devices showing that
the majority of embolic events do not occur proximal to episodes of AF
have raised doubts regarding the causal association between covert AF
and ESUS (14).
•
The authors of the ESUS construct acknowledged that arteriogenic
embolism due to non-stenotic plaques was possible in some patients with
ESUS (2).
However, grouping them into the same category as patients with other
minor cardioembolic strokes, assuming that they would also benefit from
anticoagulants, risks neglecting the atherothrombotic origin in patients
with stroke with carotid atherosclerosis with stenosis lower than 50%
or with aortic arch atheroma (AAA); these etiologies require a more
tailored approach to atherosclerosis to prevent not only stroke
recurrences, but also other vascular events such as myocardial
infarction.
Several
studies have reported a higher prevalence of ipsilateral carotid
plaques than contralateral carotid plaques in some ESUS cohorts (15, 16), and the global prevalence of carotid non-stenotic plaques in the ESUS Global Registry is as high as 79% (17).
Recently published data from the NAVIGATE trial have shown that up to
40% of the patients included in that trial had carotid plaques, this
being clearly more frequent on the ipsilateral side to the qualifying
stroke. Interestingly, the group of patients with carotid plaques showed
a strong tendency to higher frequency of stroke recurrences compared
with those without carotid plaques (18).
Similarly, AAA is a frequent finding in patients with ESUS (when screened), found in up to 28% in the ESUS Global registry (17)
and in the 29% of patients who had transesophageal echocardiography
(TEE) included in the NAVIGATE trial, and they showed a higher frequency
of multiterritorial infarcts in neuroimaging (19).
Therefore, for cases in which the stroke physician follows the ESUS
criteria, which does not require TEE, many symptomatic AAA cases might
be missed. Although clinical trials on the use of antithrombotic drugs
in stroke prevention in AAA were inconclusive because of insufficient
power calculation (19–21), these patients carry a higher risk of stroke recurrences than patients with other possible causes of cryptogenic stroke (20) and have a high burden of vascular risk factors and of coronary artery disease (19–21).
Therefore, they should be instructed to strictly adhere to lifestyle
modifications and risk factor interventions to reduce the overall
vascular risk, instead of providing them with the uncertainty of an ESUS
diagnosis.
• Patent foramen ovale (PFO) has also been included in the broad concept of ESUS (2),
following the historical controversy on its pathogenic role in ischemic
stroke. It has been reported to be present in up to 7.4% of patients
with ESUS recruited in the NAVIGATE trial (22) and in 12.6% in the RESPECT-ESUS trial (3);
however, actual rates could be underestimated, given TEE, or bubble
transcranial Doppler were not required prior to inclusion in the trials.
None of them showed NOAC to have any significant effect on reducing
stroke recurrences. Moreover, given percutaneous PFO closure has been
demonstrated to be safe and efficacious in the prevention of stroke
recurrences in those patients with ischemic stroke related to large PFOs
(especially when associated with atrial septal aneurysm) and no other
cause of stroke (23, 24),
they should be excluded from the ESUS category because the therapeutic
approach is clearly different. Indeed, an update of current nomenclature
and classifications systems has recently been proposed to include the
specific category of PFO-associated stroke (25).
• Cancer-associated stroke is another possible underlying etiology in patients with ESUS (2)
and data from the NAVIGATE trial reported a cancer diagnosis in up to
7.5% of the included patients. This value is probably an underestimate,
given an exclusion criterion was a life expectancy of <6 months. A
new cancer diagnosis at 11 months' follow-up was found in 1.7% (26).
Patients with cancer had a higher risk of stroke recurrences than
patients without cancer, without differences in ischemic stroke
recurrences between the aspirin and rivaroxaban groups, although with a
trend toward more major bleeds with rivaroxaban (26).
Involved pathogenic mechanisms are non-bacterial thrombotic
endocarditis, tumor emboli from occult cancer and a cancer-associated
hypercoagulable state. Cancer-associated stroke has a very poor
prognosis, with high mortality at follow-up (27, 28).
Identifying and treating the underlying cancer is crucial in these
patients. For this reason, following such a basic diagnostic approach as
required for ESUS appears to be inappropriate for the detection of
stroke-associated cancer and, in our opinion, further laboratory tests
(such as D-dimer, which has been proposed as a helpful parameter for
suspected covert cancer in stroke patients) (29) and cardiac examinations (such as TEE to rule out non-bacterial thrombotic endocarditis) should be performed.
•
Finally, there are some other less recognized cardioembolic sources of
stroke, such as atrial cardiopathy and left ventricular disease,
including hypertrophy, decreased ejection fraction and valvular heart
disease without AF, which merit identification in patients with stroke.
The results of the ongoing ATTICUS and ARCADIA trials (30, 31)
that are investigating the efficacy and safety of apixaban in patients
with disease of unknown etiology and atrial cardiopathy or at least one
risk factor suggestive of cardiac embolism should provide us with new
insights into the role of atrial cardiopathy and the risk of stroke,
whether mediated or not by covert AF. These trials are selecting
cryptogenic stroke patients who present the following markers suggestive
of atrial cardiopathy: left atrium enlargement >45 mm, spontaneous
echo contrast in left atrial appendage (LAA), LAA flow velocity ≤ 0.2
m/sg, atrial high rate episodes, PFO and high CHADS2-VASc score (≥4) in
the ATTICUS Trial; and the P-wave terminal force > 5,000 μV x
ms in ECG lead V1, serum N-terminal pro-brain natriuretic peptide
(NT-ProBNP) >250 pg/ml, and left atrial diameter index ≥3 cm/m2 in the ARCADIA Trial (30, 31).
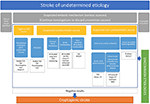
Therefore, in our opinion, the exclusion of ipsilateral
non-stenotic carotid plaques, aortic arch atherosclerosis, PFO, and
cancer-associated strokes should be a prerequisite before diagnosing a
cryptogenic stroke in clinical practice (Figure 1),
and patients with factors that have been identified as being associated
with a higher risk of covert AF should undergo long-term cardiac
monitoring. In this sense, the evaluation of serum (Nt-ProBNP) could
help in the selection of patients for long-term cardiac monitoring since
levels ≥505 pg/ml have recently shown to have a 86% sensitivity and 98%
negative predictive value for AF in cryptogenic stroke (32).
In conclusion, our advice to
stroke physicians is to forget ESUS and be smart in the search for
underlying causes of ischemic stroke, optimizing advanced diagnostic
procedures according to the patient's and stroke's characteristics,
attempting to find the correct diagnosis for stroke patients and
reducing rates of cryptogenic stroke diagnosis.

 Blanca Fuentes
Blanca Fuentes Raquel Gutiérrez-Zúñiga
Raquel Gutiérrez-Zúñiga Exuperio Díez-Tejedor
Exuperio Díez-Tejedor