Use the labels in the right column to find what you want. Or you can go thru them one by one, there are only 32,925 posts. Searching is done in the search box in upper left corner. I blog on anything to do with stroke. DO NOT DO ANYTHING SUGGESTED HERE AS I AM NOT MEDICALLY TRAINED, YOUR DOCTOR IS, LISTEN TO THEM. BUT I BET THEY DON'T KNOW HOW TO GET YOU 100% RECOVERED. I DON'T EITHER BUT HAVE PLENTY OF QUESTIONS FOR YOUR DOCTOR TO ANSWER.
Changing stroke rehab and research worldwide now.Time is Brain! trillions and trillions of neurons that DIE each day because there are NO effective hyperacute therapies besides tPA(only 12% effective). I have 523 posts on hyperacute therapy, enough for researchers to spend decades proving them out. These are my personal ideas and blog on stroke rehabilitation and stroke research. Do not attempt any of these without checking with your medical provider. Unless you join me in agitating, when you need these therapies they won't be there.
What this blog is for:
My blog is not to help survivors recover, it is to have the 10 million yearly stroke survivors light fires underneath their doctors, stroke hospitals and stroke researchers to get stroke solved. 100% recovery. The stroke medical world is completely failing at that goal, they don't even have it as a goal. Shortly after getting out of the hospital and getting NO information on the process or protocols of stroke rehabilitation and recovery I started searching on the internet and found that no other survivor received useful information. This is an attempt to cover all stroke rehabilitation information that should be readily available to survivors so they can talk with informed knowledge to their medical staff. It lays out what needs to be done to get stroke survivors closer to 100% recovery. It's quite disgusting that this information is not available from every stroke association and doctors group.
Showing posts with label anti-platelets. Show all posts
Showing posts with label anti-platelets. Show all posts
Thursday, March 9, 2017
Weighing Your Options Anticoagulants from National Stroke Association
In case you were wondering why aspirin wasn't mentioned.
Thursday, November 22, 2012
Tirofiban effectively prevents strokes in high risk patients
See if your doctor thinks this may be useful for your prevention. At least find out if you are considered high risk.
http://www.news-medical.net/news/20121122/Tirofiban-effectively-prevents-strokes-in-high-risk-patients.aspx
Scientists may have discovered a new way to prevent strokes in high risk patients, according to research from the University of Warwick and University Hospitals Coventry and Warwickshire (UHCW).
Work by a new research group, led by Professor Donald Singer, Professor of Therapeutics at Warwick Medical School and Professor Chris Imray from UHCW, has now been published in US journal Stroke.
The group is using ultrasound scanning to look at patients with carotid artery disease, one of the major causes of stroke. Clots can form on diseased carotid arteries in the neck. Small parts of these clots can released to form microemboli, which can travel to block key brain arteries and lead to weakness, disturbed speech, loss of vision and other serious stroke syndromes. Standard anti-platelet drugs such as aspirin may not prevent the formation of harmful microemboli.
The scanning process can be used to find patients at very high risk of stroke because microemboli have formed despite prior anti-platelet drugs. Using scanning, the team has found that tirofiban, another anti-platelet drug designed to inhibit the formation of blood clots, can suppress microemboli where previous treatment such as aspirin has been ineffective. In their study, tirofiban was more effective than other 'rescue' treatment.
Professor Singer said: "These findings show that the choice of rescue medicine is very important when carotid patients develop microemboli despite previous treatment with powerful anti-platelet drugs such as aspirin. We now need to go on to further studies of anti-microemboli rescue treatments, to aim for the right balance between protection and risk for our patients."
Professor Imray said: "These findings show the importance of ultrasound testing for micro-emboli in carotid disease patients. These biomarkers of high stroke risk cannot be predicted just from assessing the severity of risk factors such as smoking history, cholesterol and blood pressure."
http://www.news-medical.net/news/20121122/Tirofiban-effectively-prevents-strokes-in-high-risk-patients.aspx
Scientists may have discovered a new way to prevent strokes in high risk patients, according to research from the University of Warwick and University Hospitals Coventry and Warwickshire (UHCW).
Work by a new research group, led by Professor Donald Singer, Professor of Therapeutics at Warwick Medical School and Professor Chris Imray from UHCW, has now been published in US journal Stroke.
The group is using ultrasound scanning to look at patients with carotid artery disease, one of the major causes of stroke. Clots can form on diseased carotid arteries in the neck. Small parts of these clots can released to form microemboli, which can travel to block key brain arteries and lead to weakness, disturbed speech, loss of vision and other serious stroke syndromes. Standard anti-platelet drugs such as aspirin may not prevent the formation of harmful microemboli.
The scanning process can be used to find patients at very high risk of stroke because microemboli have formed despite prior anti-platelet drugs. Using scanning, the team has found that tirofiban, another anti-platelet drug designed to inhibit the formation of blood clots, can suppress microemboli where previous treatment such as aspirin has been ineffective. In their study, tirofiban was more effective than other 'rescue' treatment.
Professor Singer said: "These findings show that the choice of rescue medicine is very important when carotid patients develop microemboli despite previous treatment with powerful anti-platelet drugs such as aspirin. We now need to go on to further studies of anti-microemboli rescue treatments, to aim for the right balance between protection and risk for our patients."
Professor Imray said: "These findings show the importance of ultrasound testing for micro-emboli in carotid disease patients. These biomarkers of high stroke risk cannot be predicted just from assessing the severity of risk factors such as smoking history, cholesterol and blood pressure."
Tuesday, March 27, 2012
A Patient-Centered Approach to Exploring the Gastrointestinal Issues of Antiplatelet Therapy
This is Continuing Medical Education so you know at least as much as your doctor. Clopidogrel and aspirin are some of the anti-platelet therapies out there. You can sign up without being a doctor for free.
http://www.medscape.org/viewarticle/760409
Introduction
People with coronary stents are among the many cardiovascular patients prescribed long-term antiplatelet therapy. Dual antiplatelet therapy, comprising a P2Y12 inhibitor (usually clopidogrel) and aspirin, is recommended for a minimum of 12 months after drug-eluting stent placement.[1] Premature cessation of dual antiplatelet therapy in patients after acute coronary syndrome (ACS) and/or those treated with drug-eluting stents is associated with an increased risk for cardiovascular events, including stent thrombosis which can be fatal.[2]
Although this antiplatelet combination reduces the risk for ischemic events compared with aspirin alone, it is also associated with significantly more major bleeding.[3] In the VALsartan In Acute myocardial iNfarcTion trial (VALIANT) study of almost 15,000 survivors of myocardial infarction, serious gastrointestinal bleeding occurred in 0.7% over a median 2 years. Use of dual antiplatelet therapy was the most powerful predictor of this adverse event and serious gastrointestinal (GI) bleeding was associated with an increased risk of death [adjusted hazard ratio 2.54 (95% CI; 1.66-3.89)].[4] Smaller bleeds occur more frequently and long-term antiplatelet therapy is also associated with dyspepsia and other forms of GI distress.[5] Thus, proton pump inhibitors (PPIs) are often used in patients on long-term antiplatelet therapy.
Pharmacodynamic and Pharmacokinetic Interactions
Clopidogrel is a prodrug that is metabolized to its active form in a 2-step process through the cytochrome P450 (CYP) pathway with the majority of it being hydrolyzed to an inactive derivative. The CYP2C19 isoenzyme is involved in both steps (Figure 1). Most PPIs available in the United States are also metabolized via the CYP system.
http://www.medscape.org/viewarticle/760409
Introduction
People with coronary stents are among the many cardiovascular patients prescribed long-term antiplatelet therapy. Dual antiplatelet therapy, comprising a P2Y12 inhibitor (usually clopidogrel) and aspirin, is recommended for a minimum of 12 months after drug-eluting stent placement.[1] Premature cessation of dual antiplatelet therapy in patients after acute coronary syndrome (ACS) and/or those treated with drug-eluting stents is associated with an increased risk for cardiovascular events, including stent thrombosis which can be fatal.[2]
Although this antiplatelet combination reduces the risk for ischemic events compared with aspirin alone, it is also associated with significantly more major bleeding.[3] In the VALsartan In Acute myocardial iNfarcTion trial (VALIANT) study of almost 15,000 survivors of myocardial infarction, serious gastrointestinal bleeding occurred in 0.7% over a median 2 years. Use of dual antiplatelet therapy was the most powerful predictor of this adverse event and serious gastrointestinal (GI) bleeding was associated with an increased risk of death [adjusted hazard ratio 2.54 (95% CI; 1.66-3.89)].[4] Smaller bleeds occur more frequently and long-term antiplatelet therapy is also associated with dyspepsia and other forms of GI distress.[5] Thus, proton pump inhibitors (PPIs) are often used in patients on long-term antiplatelet therapy.
Pharmacodynamic and Pharmacokinetic Interactions
Clopidogrel is a prodrug that is metabolized to its active form in a 2-step process through the cytochrome P450 (CYP) pathway with the majority of it being hydrolyzed to an inactive derivative. The CYP2C19 isoenzyme is involved in both steps (Figure 1). Most PPIs available in the United States are also metabolized via the CYP system.
View abbreviations used in this activity.
Please read the introduction, then watch Dr Cryer's video commentary that includes a patient case simulation.
Introduction
People with coronary stents are among the many cardiovascular patients prescribed long-term antiplatelet therapy. Dual antiplatelet therapy, comprising a P2Y12 inhibitor (usually clopidogrel) and aspirin, is recommended for a minimum of 12 months after drug-eluting stent placement.[1] Premature cessation of dual antiplatelet therapy in patients after acute coronary syndrome (ACS) and/or those treated with drug-eluting stents is associated with an increased risk for cardiovascular events, including stent thrombosis which can be fatal.[2]
Although this antiplatelet combination reduces the risk for ischemic events compared with aspirin alone, it is also associated with significantly more major bleeding.[3] In the VALsartan In Acute myocardial iNfarcTion trial (VALIANT) study of almost 15,000 survivors of myocardial infarction, serious gastrointestinal bleeding occurred in 0.7% over a median 2 years. Use of dual antiplatelet therapy was the most powerful predictor of this adverse event and serious gastrointestinal (GI) bleeding was associated with an increased risk of death [adjusted hazard ratio 2.54 (95% CI; 1.66-3.89)].[4] Smaller bleeds occur more frequently and long-term antiplatelet therapy is also associated with dyspepsia and other forms of GI distress.[5] Thus, proton pump inhibitors (PPIs) are often used in patients on long-term antiplatelet therapy.
Pharmacodynamic and Pharmacokinetic Interactions
Clopidogrel is a prodrug that is metabolized to its active form in a 2-step process through the cytochrome P450 (CYP) pathway with the majority of it being hydrolyzed to an inactive derivative. The CYP2C19 isoenzyme is involved in both steps (Figure 1). Most PPIs available in the United States are also metabolized via the CYP system.
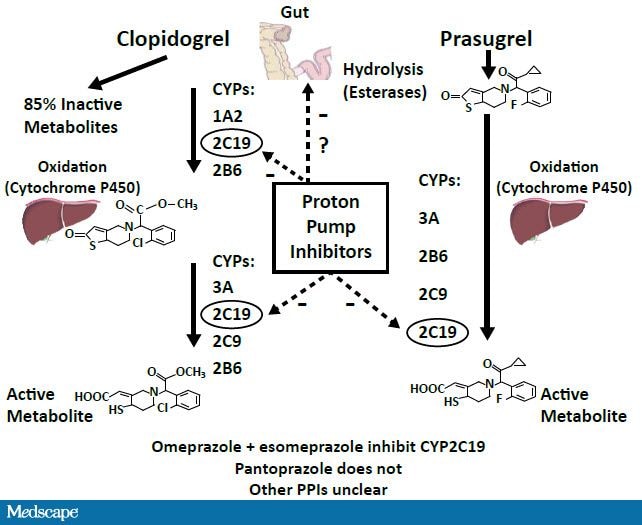 In vitro studies using assays to measure metabolite levels or platelet reactivity show that co-administration of PPIs and clopidogrel results in reduced active metabolite generation and weak inhibition of platelet reactivity.[6-7]
In vitro studies using assays to measure metabolite levels or platelet reactivity show that co-administration of PPIs and clopidogrel results in reduced active metabolite generation and weak inhibition of platelet reactivity.[6-7]
This interaction persists with spaced administration of delayed-release omeprazole and clopidogrel.[8] However, spaced administration (10 hours) of an investigational drug combining enteric-coated delayed-release aspirin (325 mg) with immediate release omeprazole (40 mg) mitigated any pharmacodynamic interaction with clopidogrel.[9] It is not clear whether these pharmacodynamic and pharmacokinetic interactions hamper the clinical efficacy of clopidogrel.
Clinical Events
Observational studies show a higher risk for cardiovascular events in clopidogrel-treated patients co-administered PPIs, notably omeprazole.[10] Large randomized studies comparing clopidogrel with newer P2Y12 inhibitors, prasugrel in the TRITON-TIMI 38 (Trials to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel -- Thrombolysis in Myocardial Infarction) trial or ticagrelor in the PLATO (PLATelet inhibition and patient Outcomes) trial have yielded conflicting findings. The TRITON-TIMI 38 study did not show higher event rates in patients assigned clopidogrel who were taking PPIs compared with those on clopidogrel and not taking PPIs, nor did it show any effect in a similar analysis of patients assigned prasugrel.[11] However, in the PLATO trial, patients on PPIs in both the clopidogrel and ticagrelor groups had worse outcomes compared with their counterparts who were not taking PPIs.[12] That any interaction was seen in the ticagrelor-treated group was surprising since ticagrelor is a direct acting P2Y12 inhibitor that does not require biotransformation. PPI use was not randomized in these trials and patients on these gastroprotective drugs tended to be higher risk at baseline, confounding the data.
To date, the Clopidogrel and Optimization of Gastrointestinal Events (COGENT) study is the only antiplatelet trial to randomize PPI use. Almost 3800 patients with an indication for dual antiplatelet therapy were randomized to a novel clopidogrel/omeprazole combination pill or clopidogrel alone; all patients were taking aspirin. The COGENT trial planned to enroll 5,000 patients but it was stopped early because the supporter and manufacturer of the clopidogrel/omeprazole pill filed for liquidation. The trial showed no difference in ischemic events between the 2 groups; indeed, those taking PPIs experienced significantly fewer upper GI bleeding events.[13]
Please read the introduction, then watch Dr Cryer's video commentary that includes a patient case simulation.
Introduction
People with coronary stents are among the many cardiovascular patients prescribed long-term antiplatelet therapy. Dual antiplatelet therapy, comprising a P2Y12 inhibitor (usually clopidogrel) and aspirin, is recommended for a minimum of 12 months after drug-eluting stent placement.[1] Premature cessation of dual antiplatelet therapy in patients after acute coronary syndrome (ACS) and/or those treated with drug-eluting stents is associated with an increased risk for cardiovascular events, including stent thrombosis which can be fatal.[2]
Although this antiplatelet combination reduces the risk for ischemic events compared with aspirin alone, it is also associated with significantly more major bleeding.[3] In the VALsartan In Acute myocardial iNfarcTion trial (VALIANT) study of almost 15,000 survivors of myocardial infarction, serious gastrointestinal bleeding occurred in 0.7% over a median 2 years. Use of dual antiplatelet therapy was the most powerful predictor of this adverse event and serious gastrointestinal (GI) bleeding was associated with an increased risk of death [adjusted hazard ratio 2.54 (95% CI; 1.66-3.89)].[4] Smaller bleeds occur more frequently and long-term antiplatelet therapy is also associated with dyspepsia and other forms of GI distress.[5] Thus, proton pump inhibitors (PPIs) are often used in patients on long-term antiplatelet therapy.
Pharmacodynamic and Pharmacokinetic Interactions
Clopidogrel is a prodrug that is metabolized to its active form in a 2-step process through the cytochrome P450 (CYP) pathway with the majority of it being hydrolyzed to an inactive derivative. The CYP2C19 isoenzyme is involved in both steps (Figure 1). Most PPIs available in the United States are also metabolized via the CYP system.

This interaction persists with spaced administration of delayed-release omeprazole and clopidogrel.[8] However, spaced administration (10 hours) of an investigational drug combining enteric-coated delayed-release aspirin (325 mg) with immediate release omeprazole (40 mg) mitigated any pharmacodynamic interaction with clopidogrel.[9] It is not clear whether these pharmacodynamic and pharmacokinetic interactions hamper the clinical efficacy of clopidogrel.
Clinical Events
Observational studies show a higher risk for cardiovascular events in clopidogrel-treated patients co-administered PPIs, notably omeprazole.[10] Large randomized studies comparing clopidogrel with newer P2Y12 inhibitors, prasugrel in the TRITON-TIMI 38 (Trials to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel -- Thrombolysis in Myocardial Infarction) trial or ticagrelor in the PLATO (PLATelet inhibition and patient Outcomes) trial have yielded conflicting findings. The TRITON-TIMI 38 study did not show higher event rates in patients assigned clopidogrel who were taking PPIs compared with those on clopidogrel and not taking PPIs, nor did it show any effect in a similar analysis of patients assigned prasugrel.[11] However, in the PLATO trial, patients on PPIs in both the clopidogrel and ticagrelor groups had worse outcomes compared with their counterparts who were not taking PPIs.[12] That any interaction was seen in the ticagrelor-treated group was surprising since ticagrelor is a direct acting P2Y12 inhibitor that does not require biotransformation. PPI use was not randomized in these trials and patients on these gastroprotective drugs tended to be higher risk at baseline, confounding the data.
To date, the Clopidogrel and Optimization of Gastrointestinal Events (COGENT) study is the only antiplatelet trial to randomize PPI use. Almost 3800 patients with an indication for dual antiplatelet therapy were randomized to a novel clopidogrel/omeprazole combination pill or clopidogrel alone; all patients were taking aspirin. The COGENT trial planned to enroll 5,000 patients but it was stopped early because the supporter and manufacturer of the clopidogrel/omeprazole pill filed for liquidation. The trial showed no difference in ischemic events between the 2 groups; indeed, those taking PPIs experienced significantly fewer upper GI bleeding events.[13]
Subscribe to:
Posts (Atom)